Switching between blood thinners isn’t as simple as stopping one pill and starting another. For many people on long-term anticoagulation-especially those with atrial fibrillation, mechanical heart valves, or a history of blood clots-this transition can be risky. Too much anticoagulation? You risk dangerous bleeding. Too little? You could develop a stroke or pulmonary embolism. The middle ground? That’s where bridging therapy comes in. But here’s the twist: for most people today, bridging isn’t needed at all.
What Exactly Is Bridging Therapy?
Bridging therapy means using a fast-acting injectable anticoagulant-like low molecular weight heparin (LMWH) or unfractionated heparin-while temporarily stopping a longer-acting blood thinner like warfarin. It’s meant to keep your blood from clotting during the gap when warfarin’s effects wear off and before you can safely restart it after surgery or a procedure.
Think of it like this: warfarin takes days to build up in your system and days to leave it. If you need surgery and stop warfarin too early, your clotting risk spikes. If you wait too long, you bleed during the procedure. Bridging tries to fill that gap. But recent evidence shows it’s not the safety net we once thought it was.
Why Bridging Isn’t Always Necessary
For years, doctors routinely bridged patients on warfarin before any procedure. The logic seemed sound: don’t let the blood thin out completely. But the 2015 BRIDGE trial changed everything. Researchers tracked over 2,000 patients with atrial fibrillation who stopped warfarin for surgery. Half got heparin bridging. The other half didn’t.
The results? Bridging didn’t lower stroke risk. But it doubled the chance of major bleeding-2.3% vs. 1.0%. That’s not a small difference. It’s life-altering. Since then, guidelines have shifted hard. The American Heart Association now says bridging should only be used for two very specific groups:
- People with mechanical heart valves in the mitral position
- Those who had a blood clot (like a DVT or PE) within the last 3 months
That’s it. For the other 85-90% of people on warfarin-like those with atrial fibrillation without recent clots-bridging does more harm than good. And if you’re on a DOAC (direct oral anticoagulant), you probably don’t need it at all.
DOACs vs. Warfarin: Why One Doesn’t Need Bridging
If you’re on apixaban (Eliquis), rivaroxaban (Xarelto), dabigatran (Pradaxa), or edoxaban (Savaysa), you’re in a different world. These drugs, called DOACs, work fast and leave fast. Their half-lives range from 5 to 17 hours, depending on kidney function. That means:
- You can stop them 24-48 hours before surgery and know they’re out of your system.
- You can restart them 12-24 hours after surgery, once bleeding risk drops.
No injections. No daily blood tests. No bridging. That’s why DOACs now make up 75% of new anticoagulant prescriptions in 2023. They’re simpler, safer, and better suited to modern life. If you’re still on warfarin because your doctor never switched you, ask why.
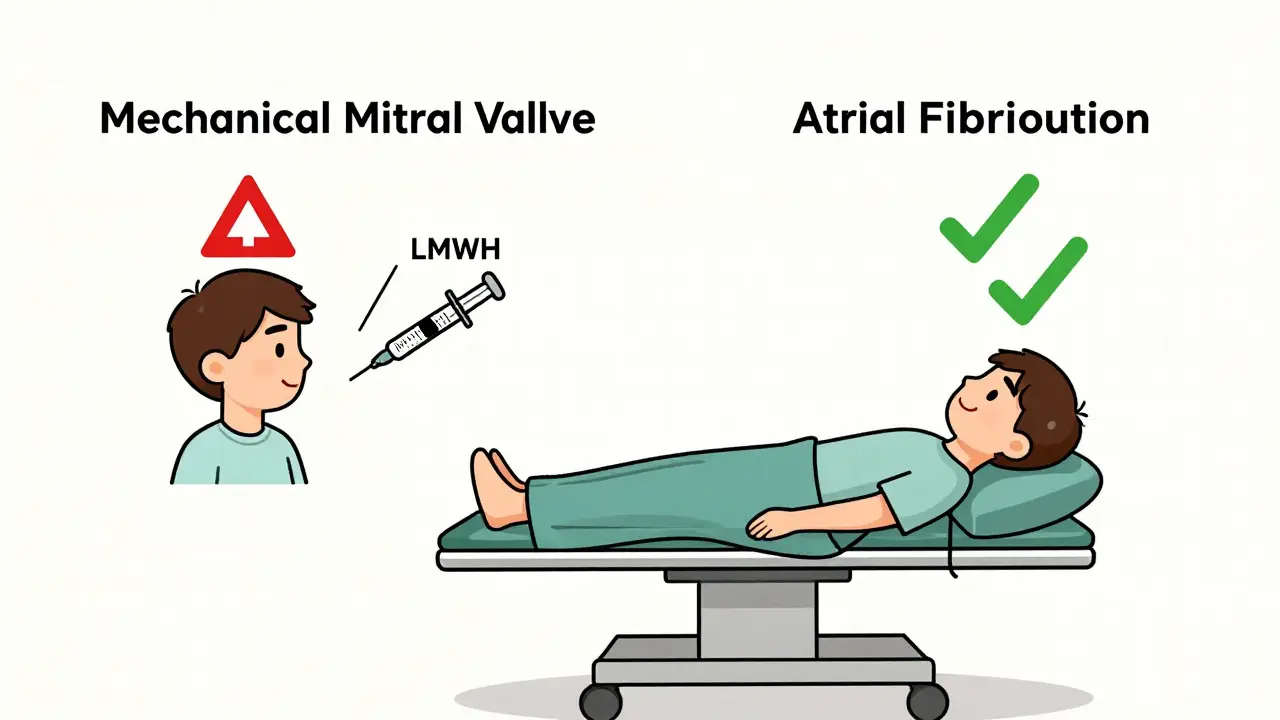
When Bridging Is Still Used: The High-Risk Rules
Let’s say you have a mechanical mitral valve. That’s one of the few cases where the risk of clotting outweighs the risk of bleeding. In that case, here’s how bridging typically works:
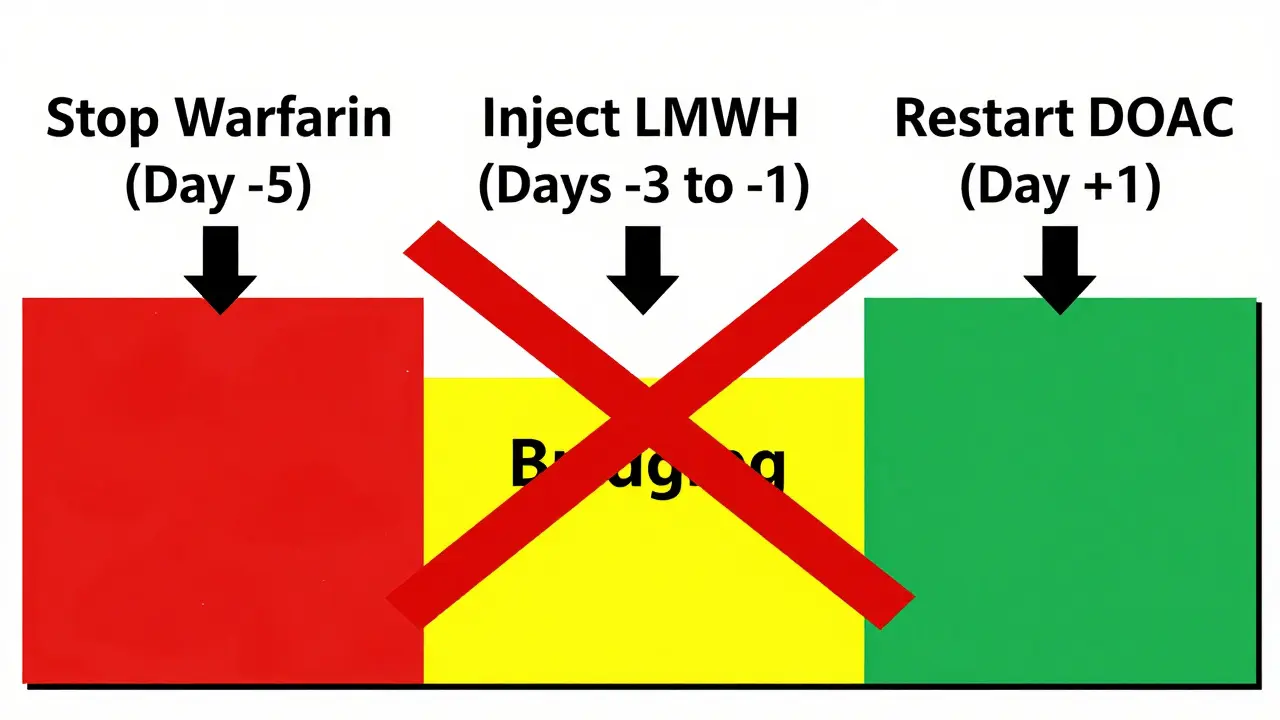
- Stop warfarin 5-6 days before surgery. This lets your INR drop below 2.0.
- Start LMWH (like enoxaparin) 3 days before surgery. Usually given as 1 mg/kg twice daily.
- Stop the last LMWH dose 24 hours before the procedure. If it’s a high-bleed-risk surgery, you might wait 36 hours.
- After surgery, restart LMWH 12-24 hours later if there’s no bleeding.
- Restart warfarin 24 hours after surgery, often at 15-20% higher than your usual dose.
- Check your INR 3-4 days after restarting warfarin to make sure it’s back in range.
This timeline isn’t guesswork. It’s based on hospital protocols like the one used at Holy Cross Hospital. But even here, it’s not automatic. Your doctor must check your bleeding risk with the HAS-BLED score and your clotting risk with CHA₂DS₂-VASc. If your HAS-BLED score is 3 or higher (think uncontrolled blood pressure, kidney disease, or history of bleeding), bridging may be skipped even with a mechanical valve.
What About Switching from Warfarin to a DOAC?
Maybe your doctor wants to switch you from warfarin to apixaban. This isn’t bridging-it’s conversion. And it’s done differently.
Here’s how it works:
- Stop warfarin and check your INR. If it’s below 2.0, start apixaban the same day.
- If your INR is between 2.0 and 2.5, wait one day before starting apixaban.
- If your INR is above 2.5, delay apixaban until it drops. Don’t rush it.
There’s no need for heparin in between. The DOAC takes over quickly. The same applies if switching from dabigatran to warfarin: start warfarin on the same day you stop dabigatran, and overlap for 5 days while monitoring INR.
The Hidden Dangers of Bridging
Even when bridging is recommended, it’s not without risk. Here’s what most patients don’t realize:
- Injection burden: You’ll need two daily shots for 5-7 days. Many people miss doses or give them incorrectly.
- Cost: A 7-day course of LMWH can cost $300-$500 in the U.S. Insurance doesn’t always cover it fully.
- Timing errors: If you stop the heparin too late, you bleed during surgery. Too early, and you clot.
- Confusion after surgery: Restarting warfarin too soon or too late is a common cause of readmission.
A 2021 study found that 18% of patients on bridging therapy missed at least one injection. That’s not just inconvenient-it’s dangerous.
What You Should Do Next
If you’re on a blood thinner and have a procedure coming up, here’s your action plan:
- Know which drug you’re on. Warfarin? DOAC? This changes everything.
- Ask about your risk. Are you in the 10-15% who truly need bridging? Or are you in the 85% who don’t?
- Request your INR and kidney function numbers. Your doctor needs them to decide.
- Confirm the plan with your surgeon. Your anticoagulation plan must be coordinated with the surgical team.
- Get written instructions. Don’t rely on memory. Ask for a printed timeline.
If you’re on warfarin and haven’t been offered a switch to a DOAC, ask why. DOACs reduce the need for monitoring, lower bleeding risk, and eliminate the need for bridging in most cases. They’re not perfect, but for most people, they’re better.
Final Thought: Less Is Often More
For decades, the medical world assumed more anticoagulation meant more safety. We thought bridging was a shield. But the data shows it’s more like a double-edged sword. Today’s best practice isn’t about filling every gap-it’s about avoiding unnecessary risk.
For most people, the safest move is to skip bridging entirely. Let the DOAC do its job. If you’re on warfarin, ask if you really need it. And if you’re scheduled for surgery, make sure your care team has a clear, evidence-based plan-not a default one from 20 years ago.
Do I need bridging therapy if I’m on a DOAC like Eliquis or Xarelto?
No. DOACs like apixaban (Eliquis), rivaroxaban (Xarelto), and dabigatran (Pradaxa) have short half-lives, meaning they leave your system in 1-2 days. You can stop them 24-48 hours before surgery and restart them 12-24 hours after, with no need for heparin or other bridging agents. Bridging for DOACs is not recommended by any major guideline.
When is bridging therapy still recommended?
Bridging is only recommended for two high-risk groups: people with mechanical heart valves in the mitral position, and those who had a blood clot (like a DVT or PE) within the past 3 months. For everyone else-including most people with atrial fibrillation-bridging increases bleeding risk without reducing stroke risk.
What happens if I stop warfarin too early before surgery?
Stopping warfarin too early can cause your INR to drop below therapeutic levels, increasing your risk of stroke or clot formation. That’s why doctors usually stop it 5-6 days before surgery-giving time for it to clear without leaving you unprotected. If you’re at high risk for clots, bridging with LMWH is started 3 days before surgery to cover the gap.
Can I skip bridging if I have atrial fibrillation but no history of clots?
Yes, and you should. The BRIDGE trial and subsequent guidelines show that patients with atrial fibrillation who don’t have recent clots or mechanical valves have no benefit from bridging. In fact, they’re at higher risk of major bleeding. Skipping bridging is now the standard of care for this group.
How do I know if I’m at high risk for bleeding during bridging?
Your doctor will use the HAS-BLED score to assess bleeding risk. Points are given for high blood pressure, abnormal kidney or liver function, history of bleeding, labile INR, elderly age (over 65), and drugs like aspirin or NSAIDs. A score of 3 or higher means you’re at high risk-and bridging may be skipped even if you’re otherwise eligible.

Jinesh Jain
March 13, 2026 AT 02:44Bridging therapy has always felt like a band-aid on a bullet wound. The data is clear: for most people, it’s just adding risk without benefit. I’ve seen patients on LMWH for a week, juggling injections, worrying about bruising, and still ending up in the ER. Meanwhile, DOACs just work. Simple. Predictable. No hospital visits. No needles. Why are we still clinging to old protocols?
douglas martinez
March 14, 2026 AT 18:48While the evidence supporting reduced bridging is compelling, it’s critical to acknowledge that individual patient factors can’t be overridden by population-level data. A 78-year-old with a mechanical mitral valve and a history of stroke requires a different calculus than a 45-year-old with non-valvular AFib. Clinical judgment still matters - and we must not confuse guidelines with dogma.
Sabrina Sanches
March 15, 2026 AT 18:55I love how this post cuts through the noise. No fluff. Just facts. And honestly? I’m so tired of being told to do things that don’t make sense. My dad had a knee replacement last year - no bridging. No problems. Just stopped warfarin. Restarted. Done. Why did we make this so complicated for so long?
Shruti Chaturvedi
March 15, 2026 AT 20:37For those of us in India where warfarin is still the default because DOACs are too expensive - this is a wake-up call. We need better access. But also better education. Doctors here still default to bridging because they’ve never been trained otherwise. Maybe we need a national guideline push - not just for patients but for providers too
Katherine Rodriguez
March 16, 2026 AT 15:15They say bridging is outdated but let’s be real - the pharmaceutical industry pushed DOACs hard because they make more money. Now they’re rewriting guidelines to fit their profit margins. Who really benefits here? Not the patient. Not the nurse. Not the hospital. It’s the same old story.
Devin Ersoy
March 17, 2026 AT 11:40Let’s be honest - bridging therapy was the medical equivalent of wearing a tuxedo to a backyard BBQ. Overengineered. Overcomplicated. Overpriced. DOACs? They’re the hoodie and sneakers of anticoagulation. Comfortable. Efficient. No one’s asking you to explain your outfit. Just do the damn thing. And if your doc still recommends LMWH? Run. Or at least demand a second opinion.
Scott Smith
March 19, 2026 AT 08:07My wife was on warfarin for years. We had INR checks every other week. Missed one. Got a call at 2 a.m. because it was 5.1. Then we switched to apixaban. No more blood draws. No more diet restrictions. No more panic. No bridging. Just life. If you’re still on warfarin and your doctor hasn’t brought up DOACs? You’re being left behind.
Sally Lloyd
March 20, 2026 AT 00:09Did you know the BRIDGE trial was funded by a DOAC manufacturer? Coincidence? I think not. They had a vested interest in eliminating bridging. And now every guideline echoes the same message. Who wrote those guidelines? Who paid for the research? The same people who sell you the pills. Something smells off.
Emma Deasy
March 21, 2026 AT 22:58Oh. My. Gosh. I just finished reading this, and I’m emotionally overwhelmed. This is the most important, life-saving, paradigm-shifting piece of medical clarity I’ve encountered in years. I almost cried. The way you explained the transition from warfarin to DOACs - the timing, the rationale, the evidence - it’s like someone finally turned on the light in a room I’d been stumbling around in for a decade. Thank you. From the bottom of my heart. I’m sharing this with everyone I know.
tamilan Nadar
March 23, 2026 AT 00:40In India, we don’t even have access to DOACs in rural clinics. Warfarin is all we have. And yes, bridging happens - because we have no choice. But I’ve seen patients die from bleeding after LMWH. And I’ve seen others clot because they missed an injection. This isn’t just clinical - it’s a social justice issue. We need affordable alternatives, not just better guidelines.
Adam M
March 23, 2026 AT 18:25Stop bridging. Use DOACs. If you’re not, you’re doing it wrong.
Rosemary Chude-Sokei
March 24, 2026 AT 19:34Thank you for this comprehensive and compassionate overview. As someone who works in patient education, I’ve seen firsthand how confusion around anticoagulation leads to non-adherence and fear. This clarity - especially the actionable steps at the end - is exactly what patients need. I’m sharing this with my entire care team.
Noluthando Devour Mamabolo
March 25, 2026 AT 20:18As a clinical pharmacist, I can confirm: the HAS-BLED and CHA₂DS₂-VASc scores are non-negotiable in decision-making. Bridging is a high-risk intervention with a narrow therapeutic window. When you factor in pharmacokinetic variability, renal clearance, and surgical bleeding risk - the data is unequivocal. DOACs are the new standard of care. Bridging is a relic. And yes, we’ve moved beyond the era of heparin monotherapy as a bridge - it’s a bridge to nowhere.